Image

SafeGuard with BV
Mitigate Risk & Ensure Compliance
Leverage a best-in-class safety & hygiene program
5 step process to receive SafeGuardTM Hygiene Excellence certification
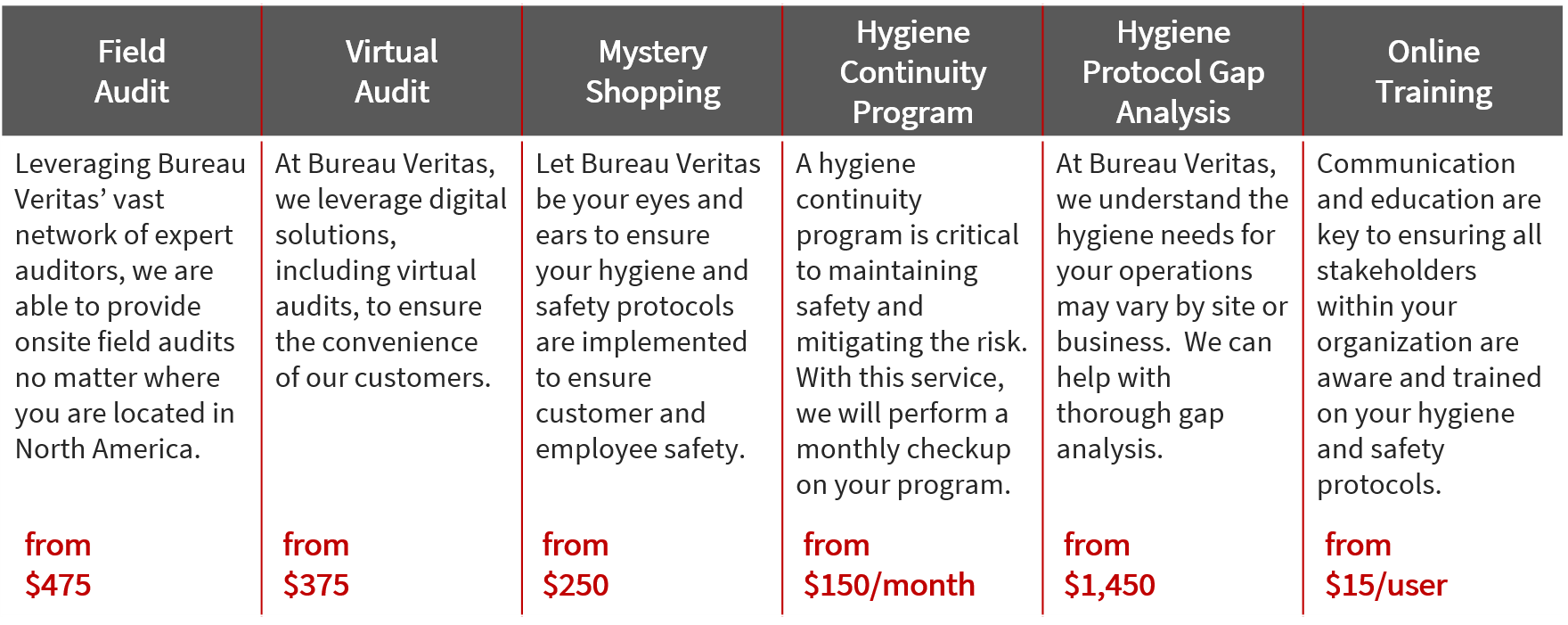
Pricing
We partner with our clients to build a custom continuity program to mitigate COVID-19 risk and ensure safety.
All prices are in USD, per location and vary based on location, audit duration, and complexity of the site/sector.
Where are you on your hygiene journey?
Based on your risk level, we will recommend which steps in the process is required for you to achieve your SafeGuardTM Hygiene Excellence and Safety Label. There are three levels of risk, High, Medium, and Low, and each risk level presents steps to move forward in your hygiene journey. Click below to find out more.
The services listed below will help you and your organization ensure the safety of your employees and customers, giving them added assurance and shaping a world of trust.
Free Download
SafeGuard Hygiene Excellence and Safety Brochure
Download Now